Back to top
About Medicine
Prescription only medicine
My Account Area
The Patient Information Leaflet (PIL) is the leaflet included in the pack with a medicine.
Last updated on emc: 30 Mar 2022
EXOCIN® 0.3% w/v
Eye drops, solution
Ofloxacin
- Keep this leaflet. You may need to read it again.
- If you have any further questions, ask your doctor or pharmacist.
- This medicine has been prescribed for you only. Do not pass it on to others. It may harm them, even if their symptoms are the same as yours.
- If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet (see section 4)
1. What EXOCIN is and what it is used for
2. What you need to know before you use EXOCIN
3. How to use EXOCIN
4. Possible side effects
5. How to store EXOCIN
6. Contents of the pack and other information
EXOCIN is an eye-drop used to treat external eye infections such as some types of conjunctivitis.
EXOCIN belongs to a group of medicines called 4-quinolone antibacterial agents.
EXOCIN is not recommended for use in infants below the age of one year.
- If you are allergic (hypersensitive) to ofloxacin, benzalkonium chloride, any of the other ingredients, or any other quinolones.
This product should be used with caution in patients sensitive to other quinolone antibacterial agents.
Long term use may result in a new bacterial infection which does not respond to EXOCIN.
This product should be used with caution in patients with a defect or ulceration of the surface of the eye.
EXOCIN potentially increases sensitivity to sunlight. You should avoid exposure to direct sunlight or sun during use of EXOCIN.
Caution should be taken when using this kind of medicine, if you were born with or have family history of prolonged QT interval (seen on ECG, electrical recording of the heart), have salt imbalance in the blood (especially low level of potassium or magnesium in the blood), have a very slow heart rhythm (called ‘bradycardia’), have a weak heart (heart failure), have a history of heart attack (myocardial infarction), you are female or elderly or you are taking other medicines that result in abnormal ECG changes (see section Taking other medicines).
There is limited experience of the use of EXOCIN in children. Talk to your doctor before you start using EXOCIN in children.
Please tell your doctor or pharmacist if you are using or have recently used any other medicine, including medicines obtained without a prescription.
You must tell your doctor if you are taking other medicines that can alter your heart rhythm: medicines that belong to the group of anti-arrhythmics (e.g. quindine, hydroquindine, disopyramide, amiodarone, sotalol, dofetilide, ibutilide), tricyclic antidepressants, some antimicrobials (that belong to the group of macrolides), some antipsychotics.
Tell your doctor before you start using EXOCIN if you are pregnant or if you are breast-feeding. Your doctor can then decide whether you can use EXOCIN.
Ask your doctor or pharmacist for advice before using any medicine.
Your sight may become blurred for a short time just after using EXOCIN. You should not drive or use machines until your sight is clear again
This medicine contains 0.05mg of benzalkonium chloride in 1 ml of solution.
Benzalkonium chloride may also cause eye irritation, especially if you have dry eyes or disorders of the cornea (the clear layer at the front of the eye). If you feel abnormal eye sensation, stinging or pain in the eye after using this medicine, talk to your doctor.
Benzalkonium chloride is a preservative which may be absorbed by soft contact lenses and may change the colour of the contact lenses. Normally you should not wear contact lenses whilst being treated with this product. However, there may be situations where use of contact lenses is unavoidable. You should remove contact lenses before using this medicine and put them back 15 minutes afterwards.
Always use EXOCIN exactly as your doctor has told you. You should check with your doctor or pharmacist if you are not sure. The usual dose is 1 or 2 drops into the affected eye(s) every 2 to 4 hours for the first 2 days and then 4 times a day from then on.
To be effective EXOCIN must be taken regularly.
The length of treatment should not exceed ten days.
You must not use the bottle if the tamper-proof seal on the bottle neck is broken before you first use it.
Apply your eye drops in the following way:
1. Wash your hands. Tilt your head back and look at the ceiling.
2. Gently pull the lower eyelid down until there is a small pocket
3. Turn the bottle upside down and squeeze it to release one or two drops into each eye that needs treatment.
4. Let go of the lower lid, and close your eye for 30 seconds
If a drop misses your eye, try again.
To avoid contamination, do not let the tip of the dropper touch your eye or anything else.
Replace and tighten the cap straight after use.
The proper application of your eye drops is very important. If you have any questions ask your doctor or pharmacist.
If you have placed too many drops in your eye(s), wash the eye(s) with clean water.
Apply your next dose at the normal time.
If, by accident, anyone drinks this medicine, contact your doctor straight away.
If you forget a dose apply it as soon as you remember, unless it is almost time for your next dose, in which case you should miss out the forgotten dose.
Then apply your next dose as usual and continue with your normal routine.
Do not use a double dose to make up for a forgotten dose.
EXOCIN should be used as advised by your doctor. If you have any further questions on the use of this product, ask your doctor or pharmacist.
Like all medicines, EXOCIN can cause side effects, but not everyone gets them.
If you have one or more of the following side effects, you may have had a serious allergic reaction. Stop taking EXOCIN immediately and contact your physician.
Allergic reactions:
Frequency not known (cannot be estimated from available data)
- Allergic reactions in the eye (including itchiness of the eye and/or eyelid)
- Inflammation of the skin due to allergy (including: rash, itching or hives)
- Severe sudden life-threatening allergic reaction (Anaphylactic) presenting as swelling beneath the skin that can occur in areas such as the face, lips or other parts of the body, swelling of the mouth, tongue or throat that can obstruct the airways which may cause wheezing, difficulty swallowing, breathing or shortness of breath
- Potentially life-threatening skin rashes (Stevens-Johnson syndrome, toxic epidermal necrolysis) have been reported with the use of EXOCIN, appearing initially as reddish target-like spots or circular patches often with central blisters on the trunk.
You should see your doctor if any of the following side effects prove troublesome or if they are long-lasting.
Common side effects (may affect up to 1 in 10 people)
- Eye irritation
- Ocular discomfort
Side effects where the frequency is not known (cannot be estimated from available data)
Affecting the eye:
- Visual disturbance
- Tearing
- Inflammation
- Redness
- Sensitivity to light
- A feeling that something is in your eye
- Eye swelling
- Swelling around the eyes (including eyelid swelling)
- Eye pain
- Dryness (mild stinging or burning)
Affecting the body:
- Dizziness
- Nausea
Affecting the heart:
- Abnormal fast heart rhythm
- Life-threatening irregular heart rhythm
- Alteration of the heart rhythm (called ‘prolongation of QT interval’, seen on ECG electrical activity of the heart)
If you get any side effects, talk to your doctor or pharmacist. This includes any possible side effects not listed in this leaflet. You can also report side effects directly via:
Yellow Card Scheme
Website: www.mhra.gov.uk/yellowcard
By reporting side effects you can help provide more information on the safety of this medicine.
Keep out of the reach and sight of children.
Once you have opened the bottle do not use it longer than 28 days, even if there is solution remaining.
Do not use EXOCIN after the expiry date which is stated on the bottle label and the bottom of the carton after EXP. The expiry date refers to the last day of that month.
Do not store above 25°C.
Do not use EXOCIN if you notice the tamper-proof seal on the bottle is broken.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
- The active substance is Ofloxacin 0.3% w/v.
- The other ingredients are benzalkonium chloride, sodium chloride, sodium hydroxide (to adjust pH), hydrochloric acid (to adjust pH) and purified water
EXOCIN is an eye drop solution in a plastic bottle. Each carton contains 1 plastic bottle with a screw cap.
AbbVie Ltd.
Maidenhead
SL6 4UB
UK
Tel: 01628 561090
Allergan Pharmaceuticals Ireland
Westport
Co. Mayo
Ireland
This leaflet was last revised in February 2022
To listen to or request a copy of this leaflet in Braille, large print or audio please call, free of charge: 0800 198 5000 (UK only).
Please be ready to give the following information: Ofloxacin 0.3% reference number PL 41042/0064. This is a service provided by the Royal National Institute of Blind People.
Частота возникновения побочных эффектов оценивалась следующим образом: часто — более 1 случая на 100 назначений; нечасто — менее 1 случая на 100 назначений; редко — менее 1 случая на 1000 назначений.
Со стороны органов ЖКТ и печени: часто — тошнота, рвота, диарея (может быть симптомом энтероколита, в некоторых случаях — геморрагического), абдоминальная боль, гастралгия, метеоризм; нечасто — потеря аппетита, повышение показателей печеночных трансаминаз и/или билирубина; редко — возможно развитие холестатической желтухи, гепатита или выраженного нарушения функции печени; псевдомембранозный колит (чаще всего вызванный Clostridium difficile) — в этом случае следует немедленно прекратить прием препарата. Не следует применять препараты, снижающие перистальтику кишечника.
Со стороны нервной системы и органов чувств: часто — головная боль, головокружение, чувство усталости; нечасто — неуверенность движений, судороги, психомоторное возбуждение, фобии, повышение внутричерепного давления, спутанность сознания, нарушение равновесия, кошмарные сновидения, чувство тревоги, депрессия, галлюцинации и психотические реакции, бессонница, шаткая походка и тремор (в результате нарушения мышечной координации), нейропатия, чувство онемения и парестезии или гиперестезии, нарушение зрения, цветовосприятия, диплопия, нарушение вкусового и обонятельного восприятия (в редких случаях — временная потеря функции); редко — снижение слуха (включая потерю слуха). Если эти реакции наблюдаются у пациентов после приема первой дозы офлоксацина, следует немедленно прекратить курс терапии.
Со стороны сердечно-сосудистой системы: тахикардия и временное снижение АД; нечасто — циркуляторный коллапс (в результате быстрого снижения АД).
Со стороны системы кроветворения: редко — анемия, лейкопения (включая агранулоцитоз), тромбоцитопения, панцитопения, возможно развитие гемолитической и апластической анемии.
Со стороны мочеполовой системы (почки): нечасто — нарушение функции почек;
редко — острый интерстициальный нефрит или острая почечная недостаточность с прогрессирующим увеличением концентрации сывороточного креатинина, повышение содержания мочевины.
Аллергические реакции и действие на кожные покровы: часто — кожная сыпь, зуд; нечасто — фотодерматит, возможно развитие аллергических реакций немедленного и замедленного типа, обычно включающих кожные проявления (например экссудативная эритема, синдром Стивенса-Джонсона, синдром Лайелла и васкулиты). В редких случаях возможно развитие других признаков анафилаксии: тахикардия, лихорадка, аллергический пневмонит, аллергический нефрит, отек Квинке, бронхоспазм, диспноэ, шок, ангионевротический отек, васкулитоподобные реакции, эозинофилия (следует немедленно прекратить применение препарата); редко — эозинофильная пневмония.
Со стороны опорно-двигательного аппарата: редко — тендинит, болевые ощущения в мышцах и суставах (в редких случаях могут являться признаком рабдомиолиза), тендовагинит, разрыв сухожилия. Даже при наличии незначительных признаков воспаления сухожилия следует немедленно прекратить прием препарата.
Прочие: редко — суперинфекция, гипер- или гипогликемия, слабость, дисбактериоз кишечника, вагинит.
У некоторых пациентов (с особой предрасположенностью) применение офлоксацина может спровоцировать атаку порфирии.
За исключением очень редких случаев (включая нарушение вкуса, обоняния и слуха) побочные эффекты временные и исчезают после прекращения применения препарата.
Изменение лабораторных показателей: увеличение показателей активности ферментов — ЩФ, АСТ, АЛТ, ЛДГ.
EXOCIN is not for injection.
Safety and effectiveness in infants below the age of one year have not been established.
Serious and occasionally fatal hypersensitivity (anaphylactic/anaphylactoid) reactions, some following the first dose, have been reported in patients receiving systemic quinolones, including ofloxacin. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, angioedema (including laryngeal, pharyngeal or facial oedema), airway obstruction, dyspnoea, urticaria, and itching.
If an allergic reaction to EXOCIN occurs, discontinue the drug. Use EXOCIN with caution in patients who have exhibited sensitivities to other quinolones antibacterial agents.
When using EXOCIN the risk of rhinopharyngeal passage which can contribute to the occurrence and the diffusion of bacterial resistance should be considered. As with other anti-infectives, prolonged use may result in overgrowth of non-susceptible organisms.
If worsening infection occurs, or if clinical improvement is not noted within a reasonable period, discontinue use and institute alternative therapy.
Cardiac disorders
Caution should be taken when using fluoroquinolones, including EXOCIN in patients with known risk factors for prolongation of the QT interval such as, for example:
- congenital long QT syndrome
- concomitant use of drugs that are known to prolong the QT interval (e.g. Class IA and III anti-arrhythmics, tricyclic antidepressants, macrolides, antipsychotics)
- uncorrected electrolyte imbalance (e.g. hypokalaemia, hypomagnesaemia)
- cardiac disease (e.g. heart failure, myocardial infarction, bradycardia)
(See section 4.2 Elderly, section 4.5, section 4.8, section 4.9).
Elderly patients and women may be more sensitive to QTc-prolonging medications. Therefore, caution should be taken when using fluoroquinolones, including EXOCIN, in these populations.
Use EXOCIN with caution in patients who have exhibited sensitivities to other quinolone antibacterial agents.
Data are very limited to establish efficacy and safety of EXOCIN eye drops 0.3% in the treatment of conjunctivitis in neonates.
The use of EXOCIN eye drops in neonates with ophthalmia neonatorum caused by Neisseria gonorrhoeae or Chlamydia trachomatis is not recommended as it has not been evaluated in such patients.
Use in elderly: No comparative data are available with topical dosing in elderly versus other age groups.
Clinical and non-clinical publications have reported the occurrence of corneal perforation in patients with pre-existing corneal epithelial defect or corneal ulcer, when treated with topical fluoroquinolone antibiotics. However, significant confounding factors were involved in many of these reports, including advanced age, presence of large ulcers, concomitant ocular conditions (e.g. severe dry eye), systemic inflammatory diseases (e.g. rheumatoid arthritis), and concomitant use of ocular steroids or non-steroidal anti-inflammatory drugs. Nevertheless, it is necessary to advise caution regarding the risk of corneal perforation when using product to treat patients with corneal epithelial defects or corneal ulcers.
Corneal precipitates have been reported during treatment with topical ophthalmic ofloxacin. However, a causal relationship has not been established.
Long-term, high-dose use of other fluoroquinolones in experimental animals has caused lenticular opacities. However, this effect has not been reported in human patients, nor has it been noted following topical ophthalmic treatment with ofloxacin for up to six months in animal studies including studies in monkeys.
EXOCIN contains the preservative benzalkonium chloride which may cause ocular irritation and discolour soft contact lenses.
Sun or UV-exposition should be avoided during use of ofloxacin due to the potential for photosensitivity.
Use of contact lenses is not recommended in patients receiving treatment for an eye infection.
Эксинта — инструкция по применению
Синонимы, аналоги
Статьи
Регистрационный номер:
ЛП-007392
Торговое наименование:
Эксинта®
Международное непатентованное или группировочное наименование:
Аминобензойная кислота
Химическое наименование:
4-аминобензойная кислота
Лекарственная форма:
капли глазные
Состав препарата на 1 мл:
| Действующее вещество: | |
| Аминобензойная кислота | 0,07 мг |
| Вспомогательные вещества: | |
| Натрия хлорид | 9,0 мг |
| Натрия гидроксида раствор 1 М | до pH 3,5-5,0 |
| Вода для инъекций | до 1 мл |
Описание
Прозрачный бесцветный или желтоватый, или коричневатый раствор.
Фармакотерапевтическая группа
Противовирусное средство для местного применения.
Код ATX:
S03D
Фармакологические свойства
Фармакодинамика
Аминобензойная кислота является индуктором интерферона, оказывает антиоксидантное, иммуномодулирующее, радиопротекторное действие, ускоряет регенерацию роговицы. Препарат не обладает тератогенным, мутагенным, эмбриотоксическим действием.
Фармакокинетика
При инстилляции в конъюнктивальный мешок аминобензойная кислота быстро всасывается и проявляет терапевтическое действие.
Показания к применению
В качестве монотерапии и в составе комплексной терапии вирусных поражений глаза:
- конъюнктивиты, кератоконъюнктивита, кератоувеиты, вызываемые вирусом герпеса (herpes simplex, herpes zoster), аденовирусами;
- кератопатии инфекционного, посттравматического и послеоперационного генеза.
Противопоказания
Повышенная чувствительность к аминобензойной кислоте и вспомогательным веществам.
Применение при беременности и в период грудного вскармливания
До настоящего времени данные клинических испытаний относительно применения препарата во время беременности, лактации, а также у детей отсутствуют. Однако препарат может применяться в том случае, если предполагаемая польза превышает риск.
Способ применения и дозы
Препарат закапывают в конъюнктивальный мешок по 1-2 капли 6-8 раз в день.
После клинического выздоровления продолжать инстилляции по 2 капли 3 раза в день в течение 7 дней.
Порядок работы с флаконом:
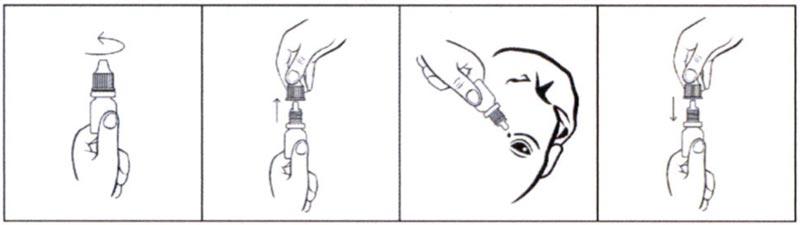
- Вращающими движениями против часовой стрелки открыть флакон и снять колпачок.
- Осторожно, не касаясь пальцами наконечника флакона, перевернуть флакон, зафиксировать между большим и указательным пальцами одной руки.
- Запрокинуть голову назад, расположить наконечник флакона над глазом и указательным пальцем другой руки оттянуть нижнее веко вниз. Слегка надавить на флакон и закапать необходимое количество препарата в конъюнктивальный мешок глаза.
Необходимо избегать контактов наконечника открытого флакона с поверхностью глаза и руками. - После использования надеть колпачок на флакон и закрыть вращающими движениями по часовой стрелке.
Порядок работы с флаконом, укомплектованным насадкой-упором для пальцев:
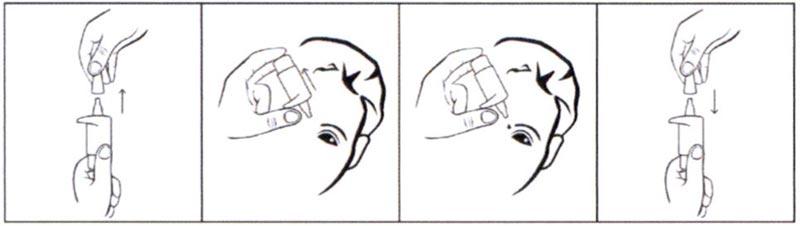
- Потянув за защитный колпачок вверх, открыть флакон.
- Осторожно, не касаясь пальцами наконечника флакона, перевернуть флакон и, расположив большой палец одной руки на специальной насадке-упоре, сделать несколько нажатий до появления первой капли.
- Запрокинуть голову назад, расположить наконечник флакона над глазом и указательным пальцем другой руки оттянуть нижнее веко вниз.
- Держа большой палец на насадке-упоре, нажать на нее и закапать необходимое количество препарата в конъюнктивальный мешок глаза.
Необходимо избегать контактов наконечника открытого флакона с поверхностью глаза и руками. - После использования надеть колпачок на флакон.
После вскрытия флакона препарат можно использовать в течение 14 дней. По истечении этого срока препарат следует выбросить.
При видимых повреждениях флакона применять препарат не следует.
Побочное действие
В очень редких случаях наблюдаются конъюнктивальная гиперемия, местные аллергические реакции.
Если любые из указанных в инструкции побочных эффектов усугубляются, или Вы заметили любые другие побочные эффекты, не указанные в инструкции, сообщите об этом врачу.
Передозировка
Данные по передозировке препарата отсутствуют.
Взаимодействие с другими лекарственными средствами
В комбинации с аналогами нуклеозидов (ацикловир, ганцикловир и др.), противомикробными средствами (фторхинолоны) и антибиотиками (полимиксин В) усиливается терапевтическое действие этих препаратов.
Если Вы применяете вышеперечисленные или другие лекарственные препараты (в том числе безрецептурные), перед применением препарата проконсультируйтесь с врачом.
Особые указания
Не применять препарат одновременно с сульфаниламидными препаратами и их производными (сульфацил натрия).
Применять препарат только согласно тем показаниям, тому способу применения и в тех дозах, которые указаны в инструкции.
Влияние на способность управлять транспортными средствами и механизмами
Не отмечено.
Форма выпуска
Капли глазные 0,007 %.
По 5 мл во флакон с капельницей из полиэтилена низкой плотности и крышкой с контролем первого вскрытия или во флакон-капельницу из полиэтилена высокого давления в комплекте с крышкой навинчиваемой и пробкой-капельницей, или во флакон из полиэтилентерефталата с капельницей пластиковой и крышкой с контролем первого вскрытия, или во флакон из коричневого стекла, укупоренный насадкой медицинской с защитным колпачком, укомплектованный насадкой-упором для пальцев или без него.
По 1 флакону из полиэтилена или полиэтилентерефталата в пакете из фольгированной пленки или без него.
По 1 флакону или по 1 пакету из фольгированной пленки с флаконом вместе с инструкцией по применению в пачке из картона.
Условия хранения
В защищенном от света месте при температуре не выше 25 °С.
Хранить в недоступном для детей месте.
Срок годности
2 года. После вскрытия флакона – 14 дней.
Не применять после окончания срока годности!
Условия отпуска
Отпускают без рецепта.
Владелец регистрационного удостоверения
ООО «Гротекс»
Россия, 195279, г. Санкт-Петербург
Индустриальный проспект, д. 71, корп. 2, лит. А
Производитель/Организация, принимающая претензии
ООО «Гротекс»
Россия, 195279, г. Санкт-Петербург
Индустриальный проспект, д. 71, корп. 2, лит. А
Купить Эксинта в ГорЗдрав
Купить Эксинта в megapteka.ru
Купить Эксинта в Планета Здоровья
*Цены в Москве. Точная цена в Вашем городе будет указана на сайте аптеки.
Комментарии
(видны только специалистам, верифицированным редакцией МЕДИ РУ)
Prescription required for this medicine
Composition
Exocin Ophthalmic Solution are primarily utilised to manage bacterial infections of the eye such as conjunctivitis and corneal ulcers. This medicine is a combination of two active ingredients and belongs to the class of antibiotic medicines.
Apart from this main use, these are also indicated for the prevention of ocular bacterial infections and serve as perioperative prophylaxis in ocular surgery. They help manage infections caused by certain strains of bacteria like Staphylococcus aureus, Streptococcus pneumoniae, Haemophilus influenzae, and Pseudomonas aeruginosa.
Before you start using this medicine, it’s vital to consult your doctor for the appropriate dosage pattern. Inform your doctor about any pre-existing conditions or medications you’re taking before starting with this. If you experience any side effects while using this medicine, let your doctor know immediately. For best results, continue using the medicine for the entire duration recommended by your doctor.
- UsesDirections for UseRoute of AdministrationSide EffectsMedicine ActivityPrecautions & WarningsInteractionsDosageStorageDiet & LifestyleFact BoxFAQs
Exocin Ophthalmic Solution are used for the following health conditions:
-
Management of bacterial conjunctivitis, commonly known as pink eye
-
Healing bacterial corneal ulcers
-
Prevention of bacterial infections in the eye
-
Prophylactic use in ocular surgery to prevent infections
Here’s how you should use Exocin Ophthalmic Solution:
-
Start with clean hands. Wash them thoroughly before you begin.
-
Tilt your head back and gently pull down your lower eyelid.
-
Carefully squeeze a few drops as prescribed by your doctor into the affected eye.
-
Close your eyes and apply gentle pressure to the inner corner to prevent the liquid from draining out.
-
Avoid touching any surfaces with the dropper tip to keep it sterile.
-
Replace the cap immediately after use.
-
If you’re using other eye medications, wait for at least 5 minutes before applying them.
Remember, these are general directions. For specific instructions, always consult your doctor.
Exocin Ophthalmic Solution are instilled topically.
Exocin Ophthalmic Solution may have the following common side effects:
-
Temporary burning, stinging, itching or discomfort upon instillation
-
Conjunctival hyperemia: This refers to a condition in which the white part of your eye, the conjunctiva, becomes red due to an increased blood flow.
-
Chemosis: This is a medical term for swollen conjunctiva, the clear tissue covering the white part of your eye and the inner surface of your eyelids.
-
Eyelid edema: This means swelling of the eyelids.
-
Foreign body sensation: You may feel like there’s something in your eye.
-
Photophobia: This is a condition where bright lights hurt your eyes.
Managing the Side Effects:
Most side effects are temporary and generally harmless, which resolve by discontinuing the use of Exocin Ophthalmic Solution. However, if you experience any serious side effects or worsening of any of the symptoms, please consult your doctor.
Here are some general guidelines for managing common side effects:
-
Temporary burning or discomfort: If this happens right after applying the drops, try closing your eyes for a few minutes after each dose.
-
Photophobia: If bright lights hurt your eyes after using Exocin Ophthalmic Solution, consider wearing sunglasses outdoors during the daytime.
Exocin Ophthalmic Solution contain two active ingredients:
-
Ofloxacin (3 mg): This fluoroquinolone antibiotic works on bacteria by inhibiting their DNA gyrase and topoisomerase IV enzymes. By disrupting these enzymes, ofloxacin interferes with bacterial DNA replication, transcription, repair and recombination processes leading to cell death.
-
Benzalkonium chloride (0.05 mg): As a quaternary ammonium compound, benzalkonium chloride acts as a preservative and bactericidal agent in Exocin Ophthalmic Solution. It disrupts bacterial cell membranes causing leakage of cellular components and eventually lysis or breakdown of the cell. It works against bacteria, fungi, protozoa, and some viruses, thus helping to prevent contamination of the multi dose container.
Pregnancy
Exercising caution is advisable while using Exocin Ophthalmic Solution during pregnancy. It’s recommended to consult your doctor before using this medicine.
Breastfeeding
If you are breastfeeding, use Exocin Ophthalmic Solution with care. Consulting with your doctor is advised.
Alcohol
Interactions of Exocin Ophthalmic Solution with alcohol are rarely seen as it is a topical medication. However, any concerns you may have should be addressed with your doctor.
Liver
For those experiencing liver health issues, caution is suggested when using Exocin Ophthalmic Solution. Your doctor can guide you based on your specific health status.
Kidney
Individuals with kidney health issues should be cautious when using Exocin Ophthalmic Solution. Consultation with your doctor is advised.
Using Machines & Driving
Temporary blurred vision may occur post the instillation of Exocin Ophthalmic Solution. Please wait until your vision clears before operating machinery or driving.
Allergy
People allergic to ofloxacin, benzalkonium chloride or any other ingredient in Exocin Ophthalmic Solution should avoid its use. Discontinue usage and consult your doctor if an allergic reaction occurs.
Children
Exocin Ophthalmic Solution can be used in children above 1 year of age under medical guidance. However, safety for children under 1 year has not been well-established. Consult your doctor before giving any medicine to your child.
Older Patients
Older patients are suggested to use Exocin Ophthalmic Solution under medical supervision due to potential susceptibility to side effects. Your doctor will guide you based on your health status.
Drug-Drug Interactions
Exocin Ophthalmic Solution interact with the following medicines:
-
Antacids, iron, zinc supplements: These can decrease the absorption of ofloxacin, an ingredient in Exocin Ophthalmic Solution. Examples include aluminium hydroxide and ferrous sulphate.
-
Oral anticoagulants: Ofloxacin may increase the effects of anticoagulants such as warfarin.
-
Medications like cyclosporine: The effects of these medications may be enhanced when used alongside Exocin Ophthalmic Solution.
Drug-Food Interactions
There are no known food interactions with Exocin Ophthalmic Solution.
Drug-Disease Interactions
Exocin Ophthalmic Solution interact with the following diseases:
-
Epithelial herpes simplex keratitis: Use Exocin Ophthalmic Solution cautiously as they may exacerbate the condition.
-
Renal impairment: Patients with kidney problems should be closely monitored due to potential increased systemic exposure to ofloxacin.
Daily Dose
The daily usage of Exocin Ophthalmic Solution should be strictly as prescribed by your doctor. The frequency and amount may vary depending on individual factors like the severity of the eye condition and how your body responds to the medicine. Make sure to abide by the prescribed dosage and do not overuse it without consulting your doctor.
Over Dose
In case you happen to put a few extra drops of this medicine in your eyes, do not panic. Rinse your eyes with clean water at room temperature. If any symptoms persist or worsen, inform your doctor.
Missed Dose
If you forget to use Exocin Ophthalmic Solution at the scheduled time, use it as soon as you remember. However, if it’s nearly time for your next dose, skip the missed dose and continue with your normal schedule. Never increase the dosage yourself without discussing it with your doctor.
-
Store Exocin Ophthalmic Solution at a temperature between 25°C-30°C.
-
Keep them away from direct sunlight and moisture. Do not freeze.
-
Use the solution within one month after opening the container.
-
Ensure the container is tightly closed when not in use.
-
Place them out of reach of children.
-
If you’re using Exocin Ophthalmic Solution for conjunctivitis, avoid touching your eyes and wash your hands frequently to prevent spreading the infection.
-
For those with corneal ulcers, avoid activities that may expose your eyes to dust, dirt, or anything that can irritate your eyes further.
Fact Box
| Available Dosage Types |
Eye Drops |
| Habit Forming |
No |
| Ailment |
Conjunctivitis, Corneal Ulcers |
| Drug Category |
DNA Gyrase Inhibitor |
| Therapeutic Category |
Antibiotic |
What are Exocin Ophthalmic Solution used for?
Exocin Ophthalmic Solution are used to manage bacterial infections of the eye, such as conjunctivitis and corneal ulcers.
How do Exocin Ophthalmic Solution work?
The drops contain ofloxacin, which kills bacteria, and benzalkonium chloride, which prevents contamination of the solution.
How should I use these eye drops?
After washing your hands, tilt your head back, pull down your lower eyelid and squeeze a few drops as prescribed by your doctor into the eye.
Can I wear contact lenses while using these drops?
You should remove contact lenses before instillation and wait 15 minutes after instilling the drops before reinserting them.
What side effects might I experience with Exocin Ophthalmic Solution?
Possible side effects include temporary burning or stinging, itching, a sensation of something in your eye, light sensitivity, or a headache. Please consult your doctor if any of this persist.
Is it safe to use these drops if I’m pregnant or breastfeeding?
Pregnant or breastfeeding women should use Exocin Ophthalmic Solution only if their doctor considers that the potential benefits outweigh the possible risks to the foetus or infant.
What should I do if I accidentally ingest this medicine?
In case of accidental oral ingestion, seek immediate medical attention. Supportive measures like stomach washing may be necessary.
Last Updated On 13 May. 2025 | 9:55AM (IST)
Manufacturer details
No.3, Kasturba Road, Level 2, Prestige Obelisk, Bangalore – 560 001., KARNATAKA, INDIA
Country of origin: INDIA
Expiry on or after: September 2025
In case of any issues, contact us:
support@truemeds.in|08069808861
Address:
6th floor, Urmi Corporate Park Solaris, Saki Vihar Road, Andheri East, Mumbai 400071
References
-
Ofloxacin: uses, interactions, mechanism of action | DrugBank Online. (n.d.). DrugBank. https://go.drugbank.com/drugs/DB01165
-
Benzalkonium: Uses, interactions, mechanism of action | DrugBank Online. (n.d.). DrugBank. https://go.drugbank.com/drugs/DB11105
-
Walimbe, T., Chelerkar, V., Bhagat, P., Joshi, A., & Raut, A. (2016). Effect of benzalkonium chloride-free latanoprost ophthalmic solution on ocular surface in patients with glaucoma. Clinical Ophthalmology, 821. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4869789/
-
U.S. National Library of Medicine. (2022). Ofloxacin Ophthalmic. MedlinePlus Drug Information. Retrieved April 1, 2024, from https://medlineplus.gov/druginfo/meds/a602029.html
-
U.S. Food and Drug Administration. (2003). Floxin (Ofloxacin). Highlights of Prescribing Information. Retrieved April 1, 2024, from https://www.accessdata.fda.gov/drugsatfda_docs/label/2003/20799slr012_floxin_lbl.pdf
Was this product description useful?
45 people find this information helpful
Disclaimer
We, at Truemeds make a diligent attempt to deliver accurate, expert- drafted and thoroughly reviewed medicine related information to our consumers. However, it should, by no means, be considered a substitute for prescription from a certified doctor or a trustworthy doctor-patient relationship. In fact we detest any form of self medication without expert advice. The sole purpose of the information on our portal is to educate our consumers, which helps improve the communication between a doctor and a patient. The information by no means is complete in its entirety and should not be used to diagnose or treat any medical condition. No amount of information can replace the experience and expertise of a doctor, therefore his/her advice always holds priority and should be followed appropriately.
